|
This episode of Medtech Snapshot features our good friend Jeff Gable, medtech embedded software SME, diving in on Agile and NPD activities. Jeff walks us through his strategy for Agile development with FDA design controls, specifically within the design input phase. Hear Jeff's 4 step approach and how a tight feedback loop is critical between requirements and system design/ architectures. #agile #embeddedsoftware #productdevelopment #medicaldevice #medtech #NPD #FDA #designcontrol #designinput #snapshot
0 Comments
Episode #20 of Medtech Snapshot features Peman Montazemi, CTO of Bright Uro - overcoming some of the biggest software development challenges for medical device applications.
#software #embeddedsystem #medicaldevice #snapshot #medtechsnapshot #medtech #softwaredevelopment #engineering #newproductdevelopment #regulation #cybersecurity #samd #spdf The world around us is ever-changing and constantly evolving, and for those of us who are leading teams, projects or companies this presents a daily challenge. One area in the business world where we commonly run into change, or scope creep as many of us like to call, is in project work. It doesn’t matter if you are running an internal project, or if you are a consultant operating externally, change in work and projects alike seems constant. If you are new to the term, scope creep is defined as “changes, continuous or uncontrolled growth, in a project’s requirements, at any point after the project begins.” Simply put, it’s a change in plans that was unplanned for after the starting point of a body of work. While complete prevention of scope creep might be unreasonable, employing strategies at the forefront of your project and throughout are vital to ensure you stay on track with as little change along the way. Strategies like the ones listed below also allow the project to ebb and flow so that some change can be accommodated while other changes, which could derail a project, are held off at the proverbial gates. Below you are some strategies to help you prevent scope creep on your next project: 1. Clearly Define the Scope Up Front Have a detailed project scope statement that outlines the project's objectives, deliverables, boundaries, and limitations. Make sure all stakeholders agree on the scope before starting the project. Special note – want to get approval quickly for your project? Make sure the project and intended outcomes align with the company’s mission and primary objectives while clearly showing a good return on investment (ROI). 2. Engage Stakeholders From the Beginning Involve key stakeholders in the scope definition and planning processes. Their input and feedback can help identify potential scope creep early on and ensure that their expectations align with the project's scope. 3. Set Realistic Goals Establish achievable project goals and objectives. Unrealistic expectations can lead to scope creep as stakeholders try to add additional features or requirements. If you can’t get agreement on the goals of the project this should cause you to pause before moving forward. Remember, each person involved may have a different perspective of what is needed based on their own biases. 4. Create a Change Control Process Develop a formalized process for requesting and approving changes to the project scope. I like to use the engineering change order (ECO) process on our projects as it formalizes changes and requires approval from stakeholders before the changes are enacted. All changes should go through this process, involving the evaluation of their impact on time, cost, and resources before being accepted. Speaking of scope creep, in this episode of Medtech Snapshot Terry Vick, Sr. Director Program Management at Shockwave Medical out of Santa Clara, CA talks about some of the strategies in this list which we need to be aware of and consider upfront in the project planning phase into order to limit scope creep on the back end. Plus, the importance of having an internal project sponsor. Back to our list of strategies to help you prevent scope creep… 5. Prioritize Requirements Use techniques like MoSCoW (Must have, Should have, Could have, Won't have), Five Whys or the Analytic Hierarchy Process (AHP) to prioritize project requirements. This helps in focusing on what's essential and avoids unnecessary additions. 6. Track Progress Against Scope Regularly track project progress against the defined scope. Tools like Microsoft Project which build out real-time Gannt charts are incredibly helpful to visualize tasks and progress. This also helps identify any deviations early on and allows for timely corrective actions. 7. Communicate to Your Team Consistently & Clearly Ensuring all team members and stakeholders understand the project's scope and the implications of scope changes is paramount to the success of the project. Effective communication can help prevent misunderstandings that might lead to scope creep. 8. Document Changes to the Project Keep a detailed record of all changes to the project scope, including their rationale and impact. This documentation helps maintain transparency and accountability. 9. Manage Expectations by Providing Project Updates to Stakeholders Continuously manage stakeholder expectations by providing regular updates on project status and any changes to the scope. This can help prevent unrealistic demands and last-minute additions. 10. Institute a Review and Approval Process for Scope Change Institute a formal review and approval process for scope changes. All changes should be evaluated by relevant stakeholders and approved based on their impact. 11. Stay Firm but Flexible While it's important to resist unnecessary scope changes, be open to valid suggestions or changes that genuinely enhance the project's value. Just ensure that these changes go through the proper evaluation and approval processes. If you can’t readily identify the ROI of the change it probably isn’t a good change to consider. 12. Conduct Regular Project Reviews Conduct regular reviews with stakeholders to ensure that the project is meeting their expectations and to address any concerns before they escalate. When facilitating meetings, try to employ Jeff Bezos’ ‘Two Pizza’ rule when it comes to meeting attendees. 13. Manage Dependencies to Avoid Unintended Cross Functional Impact Understand and manage dependencies between project tasks and deliverables. Changes in one area can often impact other parts of the project. Remember that while complete prevention of scope creep might be challenging, these strategies can significantly help to reduce its occurrence and impact on your project. Regular vigilance and proactive management are key to avoiding scope creep from taking over your project. The quickest way to overcome a business challenge is to get help from those who are experienced in besting your beast! The team at Square-1 Engineering is comprised of a variety of technical and project management professionals who are subject matter experts in the areas of NPD, Quality, Compliance and Manufacturing Engineering. Learn more about how we can solve your work and project problems today to get you back on track! This episode covers insights by Quality Executive Robert Lahaderne as he shares his perspective on the impact Root Cause makes to a medical device organization when it is done correct versus when it is done incorrectly.
#medicaldevice #medtech #rootcause In this episode we cover Medical Device Management Top Grading tips with Peter Nalbach, VP & GM, in Orange County, CA. Listen in as Pete outlines his 3-step process to align and bring in the right people to help move the company forward while simultaneously overcoming product challenges throughout the lifecycle process.
#management #medtech #medicaldevice #people #topgrading #leadership #snapshot When you’re early in the development process it can seem like there’s a never ending list of activities and projects your R&D team needs to take on. When you’re in this mode its vital R&D leaders assess time, resources and associated risks with the product they’re developing. Medical device product development executive Arif Iftekhar walks us through how to focus your team and address the most important product risks head on at an early stage. How to Solve Your #1 Business Challenge1/10/2023 One of the biggest business challenges I run across today impacting both large and small companies alike is bandwidth issues – too much work, not enough ‘resources’ to get the job done. A lack of resources in any business is a problem, indeed. Whether those resources are people, materials or relationships (supply chain), having the right number of resources to handle your needs while also being able to get a head requires both strategic forethought and action. It can be a daunting task to get beyond the fire fighting stages to then be able to actually start focusing on longer term strategic plans. For this reason its smart to get help. Despite the back and forth about the health and standing of our present economy, employment remains strong as the US unemployment rate as of December 2022 was at 3.5% according to the Bureau of Labor Statistics. For professionals in a technical setting the unemployment rate is estimated to be closer to 1.7%. This means if you need technical help most of the good resources are already in use elsewhere. When resources are constrained, yet work is plenty, this is where the consulting industry comes conveniently into play. The consulting industry has grown since 2011 by close to 5% YOY on the coattails of increasing supply and demand. With a multitude of options now available to companies looking for help one of the biggest challenges afflicting buyers seeking additional resources to solve their bandwidth issues is how they will find the right support while ensuring the money they’re paying for it results in a positive outcome. A Lesson in History Consulting isn’t new by any means, we’ve just reframed it to fit our present marketplace. Henry Ford, the USA automotive tycoon, used consultants prior to the 20th century to help build out his automotive empire ultimately creating what we know today as the Ford Motor Company. Consultants like Oliver E. Barthel are credited as key contributors to Fords success by developing combustible engines for commercial use which could be scaled for production. Needless to say consultants like Barthel and their associated contributions are immeasurable to the success of their industries. As with all things, time has a habit of bringing about change. The consulting industry is no different. One considerable difference today versus even 10 years ago is many people who are in the practice of consulting are doing so as a means to uphold a particular lifestyle. This is relatively new to the consulting game as its initial pioneers worked around the clock perfecting their art, driven by a passion to create, help and succeed on their own accord. Even as late at the early 2000s traditional consulting firms like EY, Deloitte, etc. offered excellent top notch service albeit for a hefty price. Their employees worked tirelessly to execute, the idea of a work life balance wasn’t even on the horizon. Today thousands of people go into consulting for the work flexibility. With these changes along came a fractured approach to the consulting business. The way one goes about their work (the process, focus, communication and execution) is often not the same as the next company or individual, especially when dealing with stand alone solopreneur consultants. As a result, I’ve witnessed palpable discord between consultants and their customers as an increased sensitivity between service (value and experience) and cost (time and monetary investment) unfolds. The age old discussion of value versus price isn’t new, what is new is the approach many consultants take today to justify their pricing and how they deliver their service without correlating their price to the actual value garnered by the client from the experience. I’ve witnessed, more times than I can count, consultants indicate their pricing model is based off of what is required to keep their current lifestyle in good measure, not necessarily what they’re delivering. When pricing is done without consideration of value, we may be able to gain some work in the interim, however we run the risk of leaving behind us a wake of clients who feel like they’ve been overcharged and undoubtedly under delivered. This is what I refer to as a 'consulting dilemma'. Fixing the Consulting Dilemma Pricing is important, but so is a positive outcome. While the two of these are not necessarily mutually exclusive, they do tend to have a comingled relationship. When buyers are looking for additional support price will always be a factor however it should never be the leading indicator for a decision. If you’re in a situation where you’re maxed out with your current bandwidth and you’ve determined getting consulting support is a viable option to get ahead, consider the following leading up to your buying decision:
Once you can answer these five (5) questions then you can address the pricing component of this new potential resource relationship. In order to effectively solve a customers problem a consultant must provide a service which delivers the intended results at a cost which is in alignment with the problem being solved. Long term successful consultants know and understand the importance of leaving customers feeling good about their decision to hire them for work. In fact, Salesforce, one of the largest sales CRM software companies in the world did a study with their clients where they discovered 67% of their customers said “their expectations for good experiences [with sales people] are higher than ever”. This same report revealed 76% of customers report it’s easier than ever to take their business elsewhere. This means it’s no longer about having a great tool or the best consultant qualifications, you have to provide a good experience, complete the work successfully while ensuring the price paid is in alignment with the work performed. A Key Consideration
It is indeed important to give your customers a good experience while facilitating their work, it’s just as important to price work appropriately. This is sound business advice for any professional, regardless of industry or role. A key consideration beyond experience and price, empirically important to a consultants’ success is the ability to successfully execute their work. At the end of the day if a consultant can’t successfully execute a project it doesn’t matter how great their customer service was or how affordable the price because the problem which brought the consultant to the table wasn’t solved in the first place. This is even more exacerbated in situations when a consultant charges a client using the indifference pricing model and yet still fails to successfully complete the project. When prices are high, so too are the expectations and there is often little wiggle room or understanding for anything which falls short of successful execution. When in doubt, close the project out – successfully! Conclusion To solve your business issue of to much work and not enough resources you can’t just bring in a consultant, you have to bring in the RIGHT consultant at the right price who can deliver the goods. While this may sound obvious to some, its vital consultants understand both for their future as well as reputation in the industry they serve. A Word to Consultants Davy Greenburg, a content and branding marketing consultant in Los Angeles became famous overnight in 2018 for his comment, “If I do a job in 30 minutes it’s because I spent 10 years learning how to do that job in 30 minutes. You owe me for the years, not the minutes.” When a consultant prices their services based on their ability to do such work in correlation to the problem being solved they’re much more likely to get repeat customers down the road. Lifestyle requirements and emotional decision making have no place in the process to develop your price. We’ve all experienced it – too much work, not enough time or resources to complete it. Day after day passes, the work doesn’t slow down but your timeframes become shorter and shorter furthering the stress of the looming workload. All companies, start up to conglomerate, experience this same situation. They’ve got work they can’t get to given the circumstances of their business. Usually it’s…
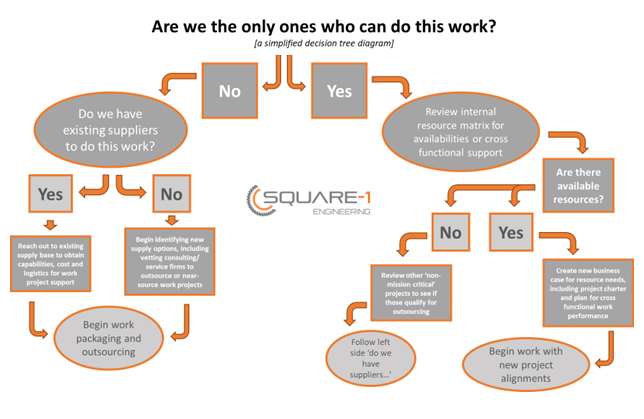
We can all agree we’ve seen this first hand and very well may be living it now within our current companies. The key question then becomes, “How do we address the ‘too much work, not enough [blank]’ commonality we all share?” First, we need to assess the work in question and is it mandatory to keep close to the chest? Meaning, are we the only ones who can do the work? If your answer is: NO – “the work can be done by someone else”; we should begin looking for alternative means for getting the work done via our supply base or strategic partners. First, review your existing base of suppliers and their capabilities to see where work can be sent out. Second, identify firms which provide outsource services. Many times the word ‘outsourcing’ is used as an all encompassing description for service providers that offer project or work package support, often which can be done onsite with the client just as easily as offsite – which would be the traditional method of outsourcing. YES – “the work can only be done by our organization”; we should review the matrix of resources versus project loads. Undoubtedly there are resources within the organization that aren’t working at full capacity and or are assigned to projects which aren’t immediate. The trick with this approach is it may not satisfy the situation long term, often times when we try to keep things in house all we do is push off the situation to a later date. If that doesn’t work, test your bias on ‘the work can only be done by us’. If you are in fact going to consider using an outsourcing or consulting firm to support you in your work projects be sure to follow this simple three step process to ensure you’re picking the right partners:
Key Take Away: Sometimes the best business decision we can make is deciding what work we want to do [internally] to increase our capacity and efficiencies while outsourcing work to suppliers or service providers which specialize in project work. In turn, this means we can focus on the mission critical work, that we enjoy and are great at doing while giving someone else the work and or projects we can’t handle or don’t want to deal with. Action Item: Utilize the below decision tree diagram (yes, it’s overly simplified) to help you determine if the work you have in question should stay internally versus would benefit from being handled by an outside source. Are you looking for an outside services firm to help you with your projects? If so, our company Square-1 Engineering, would be happy to speak with you about your needs. Check us out and let us know how we can help by clicking HERE. Solidworks: Enclosure Surfacing5/5/2022 This full length video showcases the importance of using the sweep and loft functions within Solidworks to create complex shapes. After the console and or enclosure is complete, split the model up into multiple parts to aide in proper DFM and ease of assembly.
Learn more about Square-1's CAD Services at by clicking HERE Solidworks: Sheet Metal Chassis Design4/28/2022 In this full length video we showcase our approach to chassis design using sheet metal and the importance of working smarter, not necessarily harder, by using advanced tools like Hole Wizard.
For more information on our CAD services check out CAD Services About the AuthorTravis Smith is the founder and managing director of Square-1 Engineering, a medical device consulting firm, providing end to end engineering and compliance services. He successfully served the life sciences marketplace in SoCal for over 15 years and has been recognized as a ‘40 Under 40’ honoree by the Greater Irvine Chamber of Commerce as a top leader in Orange County, CA. Categories
All
Archives
April 2024
|
Visit Square-1's
|
|







 RSS Feed
RSS Feed


