|
Medtech Snapshot presents Rodney Brenneman, medtech start-up exec, who shares his winning strategy on what is needed from a leadership perspective in order to propel a pre-clinical company forward.
Hear how emotional intelligence, adaptability and long-term strategies make the difference! If you're enjoying these Medtech Snapshots, be sure to check out our archives of past episodes HERE. #medtech #snapshot #podcast #medicaldevice #startup #entrepreneur #strategy #commercialization #emotionalintelligence #EQ #leadership
0 Comments
Medtech Snapshot Part 2:2 continues the discussion as Trisha Aure covers considerations when transitioning from 21 CFR 820 to ISO 13485.
Hear how due diligence and training play a crucial role in the transition process and the impact it has on our employees. #iso13485 #regulation #compliance #fda #21CFR #riskmanagement #medicaldevice #medtech #news #podcast #snapshot #regulatoryaffairs In this 2-part series of Medtech Snapshot we're joined by Square-1 Engineering Director of Delivery & Operations Trisha Aure as she walks us through the highlights of our article 'FDA Announcement: 21 CFR 820 and ISO 13485 Guidance'
Hear the areas where 21 CFR 820 differs most from ISO 13485 and what this means for medical device OEMs. Part-2 of this series will cover the process to transition and key considerations when doing so. #iso13485 #regulation #compliance #fda #21CFR #riskmanagement #medicaldevice #medtech #news #podcast #snapshot #regulatoryaffairs We're breaking the mold!
Medtech Snapshot returns with an enticing debate as #RAQA medtech industry experts Stephanie Rallis-Daw, RAC, CQE, CMDA and Robert Lahaderne, MBA spar on the topic of "When starting a new job/ project, what is the most common medtech compliance shortfall you can expect to encounter at your new company?" New to Medtech Snapshot? Check out our archive of past episodes at https://www.sqr1services.com/white-papers/category/snapshot covering topics in R&D, Quality, Clinical and Manufacturing. Honey for your eyes and ears, friends. #medtech #snapshot #podcast #medicaldevice #compliance #quality #regulatory #documentation #training Investing to Get Ahead at Work11/18/2022 Square-1's managing director Travis Smith is co-host of The Business Wingmen Show, a podcast covering business behaviors, strategies, leadership and much more. This week's episode...
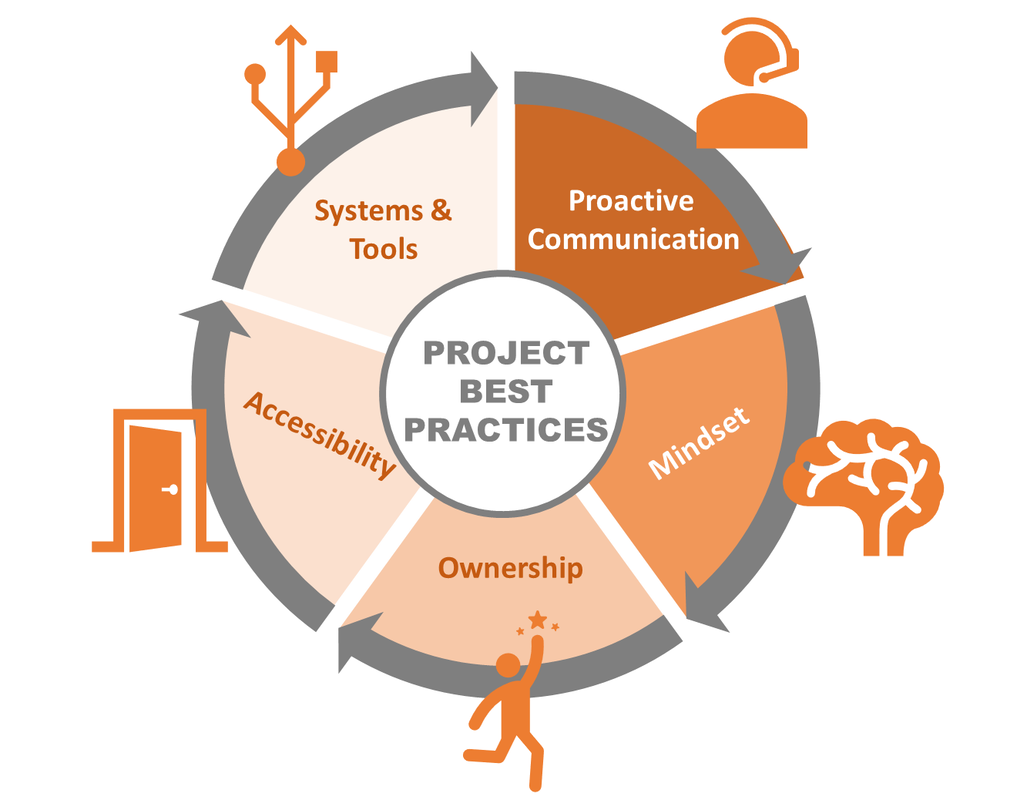
EPISODE 89: Investing to Get Ahead at Work Making advancements in your career or your business means investing in yourself and the people with potential. But, how do you know it will pay off? Listen in as your Business Wingmen podcast co-hosts, Steve Smith & Travis Smith dissect why investing to get ahead at work makes sense, and just as important doing so proactively can change the game! Listen in to episode 89 here: https://www.spreaker.com/user/businesswingmen/089-investing-to-get-ahead-at-work?utm_campaign=episode-title&utm_medium=app&utm_source=widget [This article is also featured on episode 72 of the Business Wingmen Podcast Show] Successful performance is everything! Our ability to execute at work is something we as professionals need to strive towards every day, yet many of our colleagues seem to have forgotten this time-tested reality of business. As Rory Vaden, bestselling business author and leadership speaker, accurately shares, “success is not owned, it is rented - and that rent is due everyday.” Vaden hits on a key piece which would serve all of us well to keep in mind as we come to work - our ability to successfully perform our job is what keeps us gainfully employed. I’ll take it a step further, what got us the job was our credentials, the things we’ve done in the past. What keeps us in the job thereafter is 100% our ability to get things done – the things that matter to the company and our respective performance. The same is certainly true in the consulting world, frankly I would argue it’s even more focused on performance and execution than a normal 8-5 full-time job is. The world of consulting can be summed up in 3 words: execute, execute, execute! Anything less and you’ve missed the mark. Consultants, like many of our fully employed brethren, has a tendency to forget the reason they are there, the reason they have a job – it’s to fix, solve and solution other peoples’ problems. Our inability to do this will inevitably lead to a consultant, or consulting firm, finding themselves unemployed or not getting asked back to continue supporting their customers. It’s for these reasons we need to employ the below five ‘best practices’ in our daily work. Your ability to do so will do two things – help keep you focused on what matters (successful execution of your task or job) while separating yourself from the competition and or your colleagues. Best Practice #1 – Proactive Communication If you’ve ever had some one say to you, especially someone in management, “can you give me an update on where you’re at with X”, you’ve just failed the first lesson of communication. Professionals who operate at a high level understand the importance of fluid and consistent communication. They know it’s important to communicate proactively, ahead of time. They don’t wait until the last minute to spring an urgent matter on the team or their boss, they don’t procrastinate, and they certainly don’t wait to inform those around them they are going to miss a deadline the day the deadline is due. Proactive communication is all about respecting the process and those around you. Best Practice #2 – Positive Mindset The beautiful thing about the way our minds work is we have an incredible amount of power over most of what happens in our heads. In particular, our outlook on things, our attitude, is 100% within our control. If we show up to work with a bad attitude, closed to others ideas, not welcoming feedback and or unwilling to collaborate with others we quickly will build a reputation as a person on the fringes. There’s no quicker way to alienate oneself from a team or your boss than by having a poor attitude. Yes, we all have our bad days – it happens even to the best of us. What’s important is to have your moment and then move on. Focus on what you can control and be open to ideas, different approaches and perspectives. An open and positive mindset is contagious causing your colleagues to feel invigorated working with those of us who can come to the table and leave the sourness at the door. Positive thinking also boosts our health by reducing stress! Best Practice #3 – Ownership We don’t need to be in management or the owner of a company to operate in a capacity of owning our work. Those of us who employ ‘ownership’ in our work don’t make excuses for shortcomings, we don’t worry about excuses because we’re busy finding solutions. When we own our work we don’t wait to be told what to do, we seek work proactively and or ways to improve things around us. People who operate with an ownership mentality have a tendency of being able to make decisions quicker while experiencing success more often. If you’ve ever said “that isn’t a part of my job description” you just failed the ownership best practice. People who own their work get promoted more, are typically paid higher wages and increasingly get called back to help with projects. They take responsibility for how they show up and encourage others around them to do the same. Of all the best practices listed in this article it is my humble opinion the practice of owning ones work and being accountable for it carries with it the biggest positive impact on our jobs. Best Practice #4 – Accessibility In the dawn of everyone working from home these days, or remotely, being able to get a hold of someone during normal working hours is critical. Simply put – if your boss has to track you down every time they need to reach you it probably isn’t going to end well for you in that job. Same is true if your company uses communication tools like Slack or Microsoft Teams. These tools are used to improve communication amongst the team, if you aren’t logged into the tool and or are unresponsive, you’re setting yourself up for a quick career death in your job. This doesn’t mean you need to be chained to your desk or phone eagerly awaiting the next hopeful call by a colleague or boss. What it does mean is that you need to be mindful of the various ways in which your company communicates and be attentive to those processes. If people feel like its easy to get in touch with you and or you respond quickly, they will invariably feel you are on top of things and are a reliable person to work with. For example, our company practices the ‘1/24 rule’ during standard business hours. We expect our teammates to acknowledge a communication with a response within an hour and provide an answer, or what to expect next within 24 hours. We aren’t perfect at this down to the minute (as that would be unrealistic) but I’ll be the first to say this practice makes our teammates highly proficient and therefore their jobs easier. Best Practice #5 – Systems & Tools Whether you’re a consultant, an employee or just new to your job, in today’s world there’s no excuse for not learning a company’s procedures, systems and tools. Those of us who choose not to work within these parameters typically find ourselves making careless mistakes, coming up short on our projects and irritating the heck out of our colleagues and management. Why is this? Well, when we don’t use a tool right, or don’t follow a certain SOP accurately it inevitably makes more work for someone else down the road. Trust me when I say every one hates having to fix other people’s inaccuracies, especially when it’s documentation related. If you’re new to a company do everything you can to quickly spool up by learning the tools, systems, workflows and documentation processes. The quicker you learn it the better your work product and output will be. People may just like working with you better as a result. Conclusion While there are many things which lead to someone being successful in their line of work, I firmly believe these five best practices are things all professionals can do, regardless of their experience, education level, etc. We have a choice to make every day when we show up to work – be the best at what we do or simply be average. As the job market continues to tighten up those of us who are average will find themselves unemployed far more frequently than those of us who choose to operate at a high frequency. It’s a choice – do you have what it takes? The Future of Business in California3/30/2021 TODAY AT 4 PM pacific!
Business owners of all industries and sizes have had to battle many challenges over the last year; pandemic, economic, societal, technological and political, just to name a few. All of these challenges have served up their own unique obstacles to deal with. The hosts of Business Wingmen Podcast Show Steve and Travis host a special guest, Major Williams, who will talk about today's business climate and his vision for the future of business in California. Join us to meet Major Williams - a seasoned entrepreneur, business owner and now candidate of Governor of California! Listen in on the discussion at www.businesswingmen.com #podcast #business #california #majorwilliams #economy #future #leadership #businesswingmen About the AuthorTravis Smith is the founder and managing director of Square-1 Engineering, a medical device consulting firm, providing end to end engineering and compliance services. He successfully served the life sciences marketplace in SoCal for over 15 years and has been recognized as a ‘40 Under 40’ honoree by the Greater Irvine Chamber of Commerce as a top leader in Orange County, CA. Categories
All
Archives
July 2024
|
Visit Square-1's
|
|




 RSS Feed
RSS Feed


